 Science
in Christian Perspective
Science
in Christian Perspective Science
in Christian Perspective
Science
in Christian Perspective
THE THERMODYNAMICAL TRIPLE
POINT. Implications for the Trinity
Michael J. Bozack
Surface Science Center
Department of Chemistry
University of Pittsburgh
Pittsburgh, PA
15260
From: PSCF 39 (March 1987): 39-41.
There has been little progress in the doctrine of the trinity since the Athanasian Creed and the Westminster Confession of Faith. A large number of parallel concepts have been suggested, but the use of the thermodynamical triple point as an analogy to the trinity has not been previously discussed. In this study we show that the thermodynamical triple point possesses a number of elements in common with the trinity. The triple point contains (1) a singular nature shared by three coequal but distinct subsistences, (2) economical, and (3) ontological properties. It also preserves the distinction between the classical trinity and the (4) tritheistic and (5) trimodal formulations. These rigorous requirements have been difficult to satisfy in previous analogies. The triple point provides an epistemic counterpart for our thinking about the trinity and allows for development of new perspectives.
The triple point is defined as the point where the solid,
liquid, and gaseous forms of a substance coexist in equilibrium. The concept is anticipated by the Gibbs phase rule,
according to which the largest number of phases P which can
coexist in a thermodynamical system plus the number of
degrees of freedom F equals the sum of the components C of
the system, plus 2. In algebraic terms:
P - F = C - 2
The phases P are the states of matter that make up the
system, usually solid, liquid, and gas. Each phase is homogeneous and contains bounding surfaces which allow for
mechanical separation (at least in principle). The degrees of
freedom F refer to the number of independently variable
parameters that completely specify the thermodynamic state
of the system. Such parameters are normally temperature,
pressure, and composition. The components C are the lowest
number of substances of independently variable composition
which compose the system. In a solution such as salt water,
possessing stable compounds, the number of components is
two (NaCl and H2O). In a metallic alloy, it is usually
sufficient to count the elements involved. Most pure substances possess a three-phase equilibrium point.
Application of the phase rule to a substance results in a phase diagram showing the possible phases available to the substance at varying pressures, temperatures, and compositions. The simplest phase diagram is that for a one-component system whose composition is fixed at 100% of the material under consideration. The remaining degrees of freedom (temperature and pressure) are customarily plotted on a two-dimensional graph with appropriate regions representing solid, liquid, and gas. A common example is water, whose phase diagram is reproduced in Figure 1. Equilibrium between two phases occurs along the mutual boundary of the phase regions, and equilibrium among all three phases occurs at the intersection of the regions. This intersection is the triple point.
For the case of water, which as a fixed composition, C = 1, and the maximum number of phases P of water that can coexist in equilibrium is three: ice, water, and steam. By Gibbs' phase rule:
P + F = C + 2The phase rule governs a system in equilibrium, meaning that the thermodynamic system possesses properties that are independent of time. At the triple point, equilibrium requires that rigorous control of pressure, temperature, and composition results in maintenance of the triple point indefinitely. The equilibrium is dynamic, with transitions occurring continuously between the coexisting phases, but in a way such that no apparent change is evident with the naked eye. This means that, at the triple point, boiling and condensing, melting and freezing, and subliming and freezing of gas are all going on simultaneously. For the case of water, the necessary pressure (0.006 atm.) is below I atmosphere and must be prepared in a special experimental setup.
Finally, it is noteworthy that the triple point is not equivalent to the mere existence of three forms of matter, but rather defines a unique relationship between the solid, liquid, and gaseous phases. Mere existence of three phases of matter is trimodal and therefore unsuitable for analogy to the trinity.
By analogy we mean the extension of patterns of relationship drawn from one area of experience to coordinate other
types of experience. This requires the establishment of a
number of characteristics held in common by the objects of
comparison. It is possible to identify at least seven important
areas of comparison between the triple point and the trinity.
1. Both the triple point and tke trinity possess a singular nature witk
three coequal but distinct subsistences.
The triple point and the trinity both have a singular essence and possess three subsistences which have real distinctions among them. For example, the three states of water at the triple point are conjoined by a common molecular structure, yet ice, steam, and water are quite different from one another macroscopically. The difference is manifested by the distinctive physical properties held by the states of matter, such as density, compressibility, electrical conductivity, et cetera. Because the coexisting phases at the triple point possess a distinctive set of physical properties, the union of one into three occurs without loss of identity of the phases. By comparison, the trinity is also a single essence containing three subsistences which are able to merge without loss of identity. There is an infusion of three-into-one in both models, the satisfaction of which constitutes a minimum requirement for establishment of an effective analogy to the trinity.
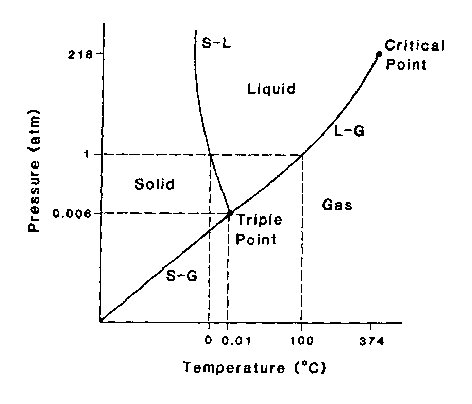 Fig. 1. Phase Diagram For Water.
The phase diagram of water, showing the possi
ble phases available to H20 at varying temperatures and
pressures. Coexistence of two phases occurs along the lines
marked S-L, L-G, and S-G, while coexistence of three phases
exists at the intersection of the regions, defined as the triple
point. Since the triple point of water occurs below atmospheric pressure, it must be put under vacuum to observe.
Fig. 1. Phase Diagram For Water.
The phase diagram of water, showing the possi
ble phases available to H20 at varying temperatures and
pressures. Coexistence of two phases occurs along the lines
marked S-L, L-G, and S-G, while coexistence of three phases
exists at the intersection of the regions, defined as the triple
point. Since the triple point of water occurs below atmospheric pressure, it must be put under vacuum to observe.
2. Both the triple point and the trinity are equilibrium
states.
Equilibrium is that condition in a thermodynamic reaction
beyond which no net change occurs in the concentration of
any substance. It defines a state in which the properties of the
system are independent of time. Dynamic equilibrium occurs
when equilibrium is maintained by equal rates of forward
and reverse reactions, as opposed to equilibrium maintained
by the absence of reaction. In the case of the triple point, the
equilibrium is dynamic. Transitions are continually occurring among the phases, and no net change in the relative
amounts of liquid, solid, and gas are measurable. The
existence of an equilibrium state is analogous to the immutability of the Godhead, meaning that the nature and attributes
of the Godhead are invariant with respect to essence. The
triple point is also invariant, since a given substance is
composed of a distinct elemental composition and, by definition, an equilibrium state is independent of time.
The Godhead is sustained by a self-contained mutuality of relations, and no one person of the trinity is or can be without the others. There is a coequal sharing of the singular divine essence without intrinsic subordination of any person. The undivided essence belongs equally to each of the persons and each possesses all the substance and all the attributes of deity. The same could be said for the triple point phases, as no state of matter is more fundamental than another, nor is water any less itself because it exists in three coincident forms.